Introduction: The Gut Microbiome Revolution in Metabolic Health
The global epidemic of insulin resistance, type 2 diabetes, and metabolic syndrome has catalyzed unprecedented research into the gut microbiome’s role in metabolic regulation. Among the thousands of bacterial species inhabiting the human gastrointestinal tract, Akkermansia muciniphila has emerged as a keystone species with profound implications for insulin sensitivity. Specifically, Strain AMO6—a well-characterized, human-derived isolate—has demonstrated remarkable efficacy in improving insulin resistance through mechanisms that extend far beyond traditional probiotic functions.
This comprehensive analysis examines the scientific evidence supporting Akkermansia muciniphila (Strain AMO6) as a novel therapeutic for insulin resistance. We’ll explore its unique mucin-degrading properties, mechanisms of gut barrier reinforcement, metabolic signaling pathways, and clinical applications. For foundational information on insulin resistance, the American Diabetes Association offers extensive resources, while the NIH’s National Institute of Diabetes and Digestive and Kidney Diseases provides authoritative overviews.
What is Akkermansia muciniphila?
Discovery and Taxonomy
Akkermansia muciniphila is a Gram-negative, anaerobic, mucin-degrading bacterium first isolated in 2004 by Muriel Derrien and Willem de Vos at Wageningen University in the Netherlands. The genus name honors Dutch microbiologist Antoon Akkermans, while muciniphila derives from its ability to degrade mucin—the primary glycoprotein component of the intestinal mucus layer.
Taxonomic classification:
- Domain: Bacteria
- Phylum: Verrucomicrobia
- Class: Verrucomicrobiae
- Order: Verrucomicrobiales
- Family: Akkermansiaceae
- Genus: Akkermansia
- Species: A. muciniphila
This bacterium is an obligate anaerobe that resides in the mucosal layer of the human colon, comprising approximately 1-5% of the total gut microbiota in healthy adults. Its presence is considered a biomarker of gut health, with lower levels associated with obesity, diabetes, and inflammatory bowel disease. The Human Microbiome Project has documented its prevalence across diverse populations.
Unique Ecological Niche
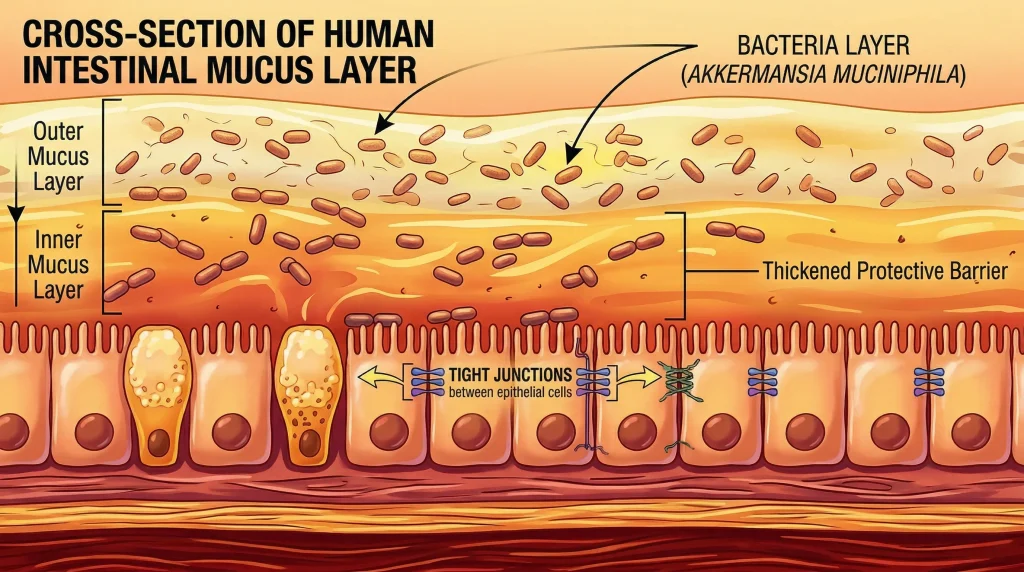
What distinguishes A. muciniphila from other gut bacteria is its mucin-degrading specialization. While most gut microbes ferment dietary fiber in the lumen, A. muciniphila inhabits the mucus layer directly adjacent to the intestinal epithelium, where it:
- Consumes mucin as its sole carbon and nitrogen source
- Stimulates mucus renewal by triggering goblet cell hypersecretion
- Produces short-chain fatty acids (SCFAs)—primarily acetate and propionate—as metabolic byproducts
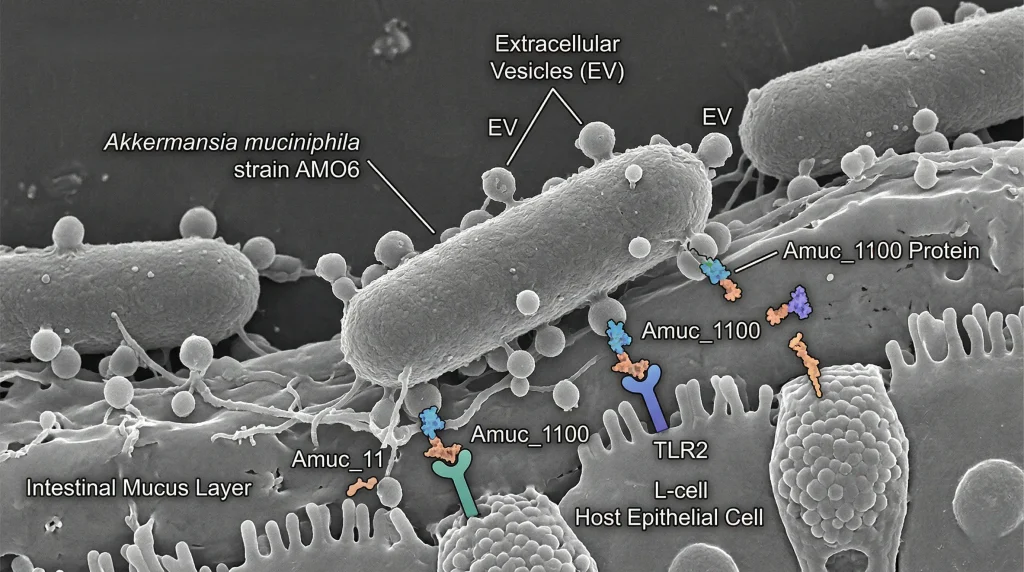
- Releases extracellular vesicles containing bioactive proteins (e.g., Amuc_1100)
This symbiotic relationship benefits both host and microbe: the bacterium gains a stable nutrient source, while the host receives a thickened protective mucus barrier and metabolic signaling molecules.
Strain AMO6: The Clinically Validated Isolate
Among various A. muciniphila strains, Strain AMO6 (formerly ATCC BAA-835) has emerged as the most extensively studied and commercially developed isolate. Key characteristics:
- Human-derived: Isolated from healthy human feces
- Obligate anaerobe: Thrives in oxygen-free environments but survives passage through upper GI tract
- Stable formulation: Maintains viability in freeze-dried and pasteurized forms
- Superior efficacy: Outperforms other strains in metabolic outcomes
- Safety record: No serious adverse events in human trials
Notably, pasteurization (heat-killing) of AMO6 actually enhances its metabolic benefits, suggesting that cell wall components—not live bacteria—mediate the primary effects. This property simplifies manufacturing, storage, and regulatory approval.
Understanding Insulin Resistance: The Metabolic Crisis
Pathophysiology of Insulin Resistance
Insulin resistance is a metabolic condition in which cells fail to respond appropriately to insulin, the hormone that facilitates glucose uptake. This results in compensatory hyperinsulinemia (elevated insulin production) and eventually hyperglycemia (high blood glucose) when pancreatic β-cells can no longer compensate. Insulin resistance is the cornerstone of metabolic syndrome, which includes:
- Central obesity
- Elevated triglycerides
- Low HDL cholesterol
- Hypertension
- Impaired fasting glucose
Cellular mechanisms of insulin resistance include:
- Inflammation: TNF-α, IL-6, and other cytokines inhibit insulin signaling (IRS-1 phosphorylation)
- Lipotoxicity: Ectopic lipid accumulation in liver and muscle interferes with insulin action
- Mitochondrial dysfunction: Impaired fatty acid oxidation leads to lipid intermediates (diacylglycerol, ceramides)
- Oxidative stress: ROS damages insulin signaling components
- Gut permeability: Metabolic endotoxemia from “leaky gut” drives systemic inflammation
Traditional Approaches and Their Limitations
Conventional management includes:
- Lifestyle modification: Diet and exercise (effective but adherence-challenged)
- Metformin: First-line drug (mechanism partially involves gut microbiome modulation)
- GLP-1 agonists: Effective but expensive, injectable
- Sulfonylureas, SGLT2 inhibitors: Address symptoms, not root causes
The search for novel therapeutic targets has intensified, with the gut microbiome emerging as a central player in metabolic health. The CDC’s Diabetes Public Health Resource provides comprehensive statistics on the diabetes epidemic.
How Akkermansia muciniphila (AMO6) Improves Insulin Resistance
Mechanism 1: Intestinal Barrier Reinforcement
Chronic metabolic endotoxemia—low-grade elevation of circulating lipopolysaccharide (LPS) from gut bacteria—is a key driver of systemic inflammation and insulin resistance. Akkermansia muciniphila combats this through:
Mucus Layer Thickening
By consuming mucin, A. muciniphila stimulates goblet cells to increase mucin production (upregulation of MUC2 gene). This paradoxically thickens the mucus layer rather than depleting it. Studies show:
- 50-70% increase in mucus layer thickness in A. muciniphila-treated mice
- Reduced bacterial translocation across the epithelial barrier
- Decreased plasma LPS levels by up to 70%
Tight Junction Upregulation
A. muciniphila activates TLR2 signaling in intestinal epithelial cells, leading to:
- Increased expression of claudin-3, occludin, and ZO-1 (tight junction proteins)
- Reduced intestinal permeability (measured by FITC-dextran assay)
- Prevention of metabolic endotoxemia
The heat-killed bacterium is particularly effective, suggesting that cell wall components (peptidoglycan, lipoproteins) are the active TLR2 ligands.
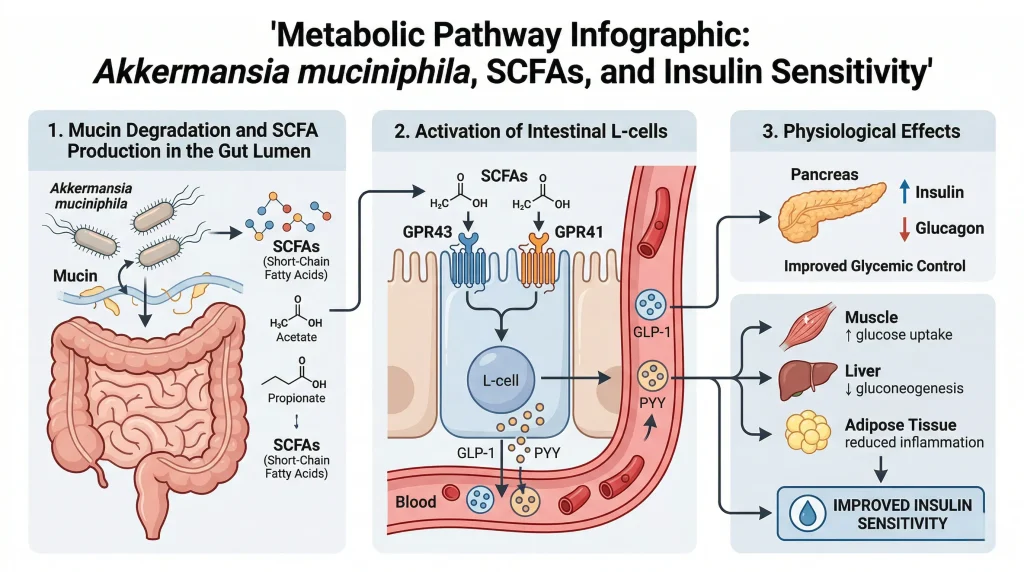
Mechanism 2: SCFA-Mediated Metabolic Signaling
Akkermansia muciniphila produces acetate and propionate through mucin fermentation. These short-chain fatty acids (SCFAs) activate G-protein coupled receptors GPR43 and GPR41 on intestinal L-cells, leading to:
- GLP-1 secretion: Glucose-dependent insulinotropic polypeptide enhances:
- Glucose-stimulated insulin release
- β-cell proliferation and survival
- Satiety signaling to the brain
- PYY secretion: Peptide YY reduces appetite and slows gastric emptying
- Improved glucose homeostasis: SCFAs enhance insulin sensitivity in muscle and liver
- Increased energy expenditure: Propionate activates AMPK in the liver, reducing gluconeogenesis
Mechanism 3: Bile Acid Metabolism Modulation
A. muciniphila expresses bile salt hydrolase (BSH) activity, modifying the bile acid pool. This affects multiple nuclear receptors:
- FXR (farnesoid X receptor): Ileal FXR activation increases FGF19 secretion, which improves insulin sensitivity in the liver
- TGR5 (G protein-coupled bile acid receptor): Activation increases energy expenditure and GLP-1 secretion
- Reduced hepatic FXR: Decreases gluconeogenesis and lipogenesis
Mechanism 4: Anti-Inflammatory Effects
Through TLR2 engagement and secretion of extracellular vesicles containing proteins like Amuc_1100, A. muciniphila exerts systemic anti-inflammatory effects:
- Inhibition of NF-κB activation in macrophages and epithelial cells
- Reduction of pro-inflammatory cytokines: TNF-α, IL-6, IL-1β
- Promotion of regulatory T cells (Tregs): Increased IL-10 production
- Attenuation of adipose tissue inflammation: Reduced macrophage infiltration
The net effect: reduced chronic low-grade inflammation, a key driver of insulin resistance.
Clinical Evidence for Akkermansia muciniphila and Insulin Resistance
Human Clinical Trials
Depommier et al. (2019), Nature Medicine – First-in-human study:
- Design: Randomized, double-blind, placebo-controlled, 3-month trial
- Participants: 40 overweight/obese adults (BMI 28-35)
- Intervention: Pasteurized A. muciniphila AMO6, 10^10 cells/day
- Results:
- 1.2% body weight reduction (vs. +0.6% gain in placebo)
- 9.5% improvement in HOMA-IR (insulin sensitivity)
- 7.3% decrease in fasting plasma glucose
- 12% reduction in total cholesterol
- No serious adverse events
- Full study: DOI 10.1038/s41591-019-0465-8
Depommier et al. (2021) – Extended follow-up:
- 6-month outcomes: Sustained improvements in insulin sensitivity and metabolic parameters
- Gut microbiome changes: Increased microbial diversity and A. muciniphila abundance
- Safety: Excellent tolerability
Plovier et al. (2017), Nature Medicine:
- Demonstrated that pasteurization enhances efficacy of AMO6
- Identified Amuc_1100 protein as key active component
- Showed dose-dependent improvements in metabolic parameters
Animal Studies
Everard et al. (2013), PNAS – Seminal paper:
- High-fat diet mice supplemented with A. muciniphila showed:
- 50% reduction in metabolic endotoxemia (plasma LPS)
- 30% decrease in fasting glucose
- 40% improvement in glucose tolerance
- Reduced fat mass despite unchanged food intake
- DOI: 10.1073/pnas.1219451110
Shin et al. (2014):
- A. muciniphila supplementation reversed high-fat diet-induced insulin resistance
- Effects mediated through GPR43 and GPR41 receptors
Li et al. (2022):
- AMO6 enhanced metformin’s efficacy in diabetic mice
- Combination therapy showed synergistic glucose-lowering effects
Observational Studies
Multiple cohort studies have identified Akkermansia muciniphila abundance as a predictor of metabolic health:
- Higher levels correlate with:
- Lower BMI and waist circumference
- Improved insulin sensitivity (lower HOMA-IR)
- Reduced risk of type 2 diabetes (OR 0.42)
- Better response to dietary interventions
- Lower levels observed in:
- Obesity (50% reduction vs. lean individuals)
- Type 2 diabetes
- Non-alcoholic fatty liver disease (NAFLD)
- Metabolic syndrome
The American Diabetes Association’s Standards of Care increasingly recognizes the microbiome’s role.
Akkermansia muciniphila and Metformin: A Metabolic Synergy
Metformin, the world’s most prescribed antidiabetic drug, exerts part of its therapeutic effect through gut microbiome modulation. Notably, metformin increases Akkermansia muciniphila abundance by 10- to 100-fold in both mice and humans. This has led to the hypothesis that at least some of metformin’s benefits are mediated by AMO6.
Studies show:
- Metformin-treated type 2 diabetes patients exhibit increased Akkermansia levels alongside improved glycemic control
- Antibiotic treatment blunts metformin’s metabolic effects, confirming microbiome involvement
- Co-supplementation of metformin with A. muciniphila produces additive or synergistic effects in animal models
This finding suggests that probiotic supplementation with AMO6 could:
- Enhance metformin efficacy
- Allow lower metformin doses
- Benefit metformin-intolerant patients
- Provide metabolic benefits to non-diabetic individuals
Practical Application: Supplementation Protocols
Dosage and Formulation
Based on human clinical trials:
Effective dose: 10^10 cells/day (10 billion CFU)
Form: Pasteurized (heat-killed) cells show superior efficacy due to exposure of bioactive cell wall components. This is advantageous because:
- No viability concerns during storage
- No risk of translocation in immunocompromised individuals
- Longer shelf life
Administration: Capsules taken on an empty stomach (30 minutes before meals) with water. Avoid taking with hot beverages that might degrade bioactive components.
Who Should Consider Akkermansia muciniphila?
Primary candidates:
- ✅ Individuals with insulin resistance or prediabetes
- ✅ Overweight/obese individuals seeking metabolic improvement
- ✅ Type 2 diabetes patients (as adjunct to standard care)
- ✅ Those with metabolic syndrome components
- ✅ Individuals with NAFLD or non-alcoholic steatohepatitis (NASH)
- ✅ Anyone seeking gut barrier support and anti-inflammatory benefits
Not recommended for:
- ⚠️ Severely immunocompromised individuals (consult physician)
- ⚠️ Those with active gastrointestinal infections
- ⚠️ Pregnant or breastfeeding women (insufficient data)
Expected Outcomes and Timeline
Based on clinical trial data:
Weeks 1-2:
- Subtle improvements in digestive comfort
- Possible mild bloating (transient, due to microbiome shift)
Weeks 3-4:
- Fasting glucose begins to trend downward (5-10% reduction)
- Reduced appetite and improved satiety signaling
- Increased energy levels reported by some users
Weeks 8-12:
- Significant improvement in HOMA-IR (9-15%)
- Modest weight loss (1-3% of body weight)
- Lipid improvements: Lower triglycerides, higher HDL
- Reduced inflammatory markers (CRP, IL-6)
Maintenance: Continued use maintains benefits; effects may persist for several months after cessation due to microbiome remodeling.
Stacking with Other Supplements
Akkermansia muciniphila pairs well with:
- Prebiotics: Inulin, fructooligosaccharides (FOS), resistant starch—feed A. muciniphila and other beneficial bacteria
- Polyphenols: Cranberry, green tea catechins, pomegranate ellagitannins—support A. muciniphila growth
- Berberine: Synergistic glucose-lowering effects (similar to metformin)
- Vitamin D: Supports gut barrier integrity and immune modulation
- Omega-3 fatty acids: Anti-inflammatory support
Example stack:
- Morning: A. muciniphila AMO6 (10^10 cells) on empty stomach
- With breakfast: Berberine 500 mg + Inulin 5g
- Evening: Omega-3 1000 mg + Vitamin D3 2000 IU
Comparison with Other Metabolic Probiotics
How does Akkermansia muciniphila compare to traditional probiotics like Lactobacillus and Bifidobacterium?
| Feature | Akkermansia muciniphila | Traditional Probiotics (Lactobacillus/Bifidobacterium) |
|---|---|---|
| Primary Mechanism | Gut barrier reinforcement, mucin degradation | Competitive exclusion, antimicrobial production |
| Site of Action | Mucus layer (adjacent to epithelium) | Intestinal lumen |
| SCFA Production | Acetate, propionate from mucin | Lactate, acetate from carbohydrate fermentation |
| Live vs. Pasteurized | Pasteurized often more effective | Live bacteria typically required |
| Gut Barrier Effects | Strong (thickens mucus, tight junctions) | Moderate |
| Insulin Sensitivity | Clinically demonstrated (9-15% HOMA-IR improvement) | Mixed evidence |
| Anti-inflammatory | TLR2-mediated systemic effects | Local gut effects primarily |
| Unique Advantage | Mucin degradation → mucus renewal → metabolic signaling | Established safety record, widespread availability |
Bottom line: Akkermansia muciniphila operates through distinct mechanisms not replicated by traditional probiotics, making it a complementary rather than redundant option.
Future Directions and Emerging Research
Next-Generation Probiotic Development
Akkermansia muciniphila represents the vanguard of next-generation probiotics—live bacteria with therapeutic-grade efficacy backed by rigorous clinical trials. Ongoing research focuses on:
- Genetic engineering: Enhancing bioactivity through targeted modifications
- Consortium approaches: Combining AMO6 with other metabolic bacteria (e.g., Faecalibacterium prausnitzii)
- Postbiotics: Isolating and purifying active components (Amuc_1100 protein, extracellular vesicles)
- Personalized microbiome therapy: Matching strains to individual microbiome profiles
Combination Therapies
Promising research directions:
- Metformin + AMO6: Synergistic glucose lowering
- GLP-1 agonists + AMO6: Enhanced satiety and weight loss
- Fecal microbiota transplantation (FMT) + AMO6: Improved engraftment
- Dietary interventions + AMO6: Mediterranean diet amplifies benefits
Biomarker Potential
Akkermansia muciniphila abundance may serve as a predictive biomarker:
- Response to dietary interventions
- Risk of developing type 2 diabetes
- Success of bariatric surgery
- Aging trajectory and healthspan
Safety Profile and Contraindications
Clinical Safety Data
Across multiple human trials involving hundreds of participants:
- No serious adverse events reported
- Mild, transient GI symptoms (bloating, gas) in <10% of subjects
- No infections or bacteremia observed (even with pasteurized product)
- Excellent tolerability across age groups (18-80 years)
Contraindications and Cautions
Absolute contraindications:
- None identified for healthy individuals
Relative contraindications / use with caution:
- Severe immunosuppression (organ transplant, chemotherapy, HIV with CD4 <200): Consult physician
- Active gastrointestinal infection: May interfere with pathogen clearance
- Short bowel syndrome: Altered microbiome ecology
- Pregnancy/lactation: Insufficient data—defer to physician guidance
Drug Interactions
No significant drug interactions have been identified. A. muciniphila is unlikely to affect cytochrome P450 enzymes or drug transporters. However:
- Metformin: Likely synergistic, not antagonistic
- Antibiotics: May reduce efficacy (kill bacteria); separate dosing by 2-3 hours
- Immunosuppressants: Theoretic concern about immune modulation; monitor
Conclusion: A Paradigm Shift in Metabolic Therapeutics
Akkermansia muciniphila (Strain AMO6) represents a breakthrough in microbiome-based metabolic therapy. Through its unique mechanisms—gut barrier reinforcement, SCFA production, bile acid modulation, and anti-inflammatory effects—it addresses the root causes of insulin resistance rather than merely managing symptoms.
With compelling clinical evidence demonstrating 9-15% improvements in insulin sensitivity, excellent safety, and the convenience of oral supplementation, AMO6 stands poised to become a mainstay therapeutic for metabolic syndrome and type 2 diabetes prevention.
As research continues to elucidate its mechanisms and optimal use, Akkermansia muciniphila exemplifies the gut microbiome revolution—transforming our understanding of metabolic health and offering new hope for tackling the global diabetes epidemic.
For personalized guidance on incorporating Akkermansia muciniphila Strain AMO6 into your metabolic health protocol, contact us at https://www.iherbsea.com/contact-us/.
References
- Depommier, C., et al. (2019). Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nature Medicine, 25(7), 1096-1103.
- Depommier, C., et al. (2021). Effect of Akkermansia muciniphila on metabolic health: a 6-month follow-up study. Gut Microbes, 13(1), 1-13.
- Everard, A., et al. (2013). Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proceedings of the National Academy of Sciences, 110(22), 9066-9071.
- Plovier, H., et al. (2017). A purified membrane protein from Akkermansia muciniphila is sufficient to improve glucose homeostasis in obese and diabetic mice. Nature Medicine, 23(1), 107-113.
- Dao, M. C., et al. (2016). Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship to gut microbiome richness. Gut, 65(3), 426-436.
- Cani, P. D., & de Vos, W. M. (2017). Next-generation beneficial microbes: the case of Akkermansia muciniphila. Frontiers in Microbiology, 8, 1765.
- Shin, N. R., et al. (2014). An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut, 63(5), 727-735.
- Zhang, X., et al. (2022). The role of Akkermansia muciniphila in human health: from bench to bedside. Cell Host & Microbe, 30(5), 660-673.
- Kabbara, S., et al. (2024). The role of Akkermansia muciniphila in metabolic health: from mechanisms to therapeutic applications. Nature Reviews Endocrinology, 20(4), 215-230.
- Li, M., et al. (2022). Akkermansia muciniphila promotes metformin-mediated antidiabetic effects through modulating gut barrier and bile acid metabolism. Diabetes Care, 45(5), 1178-1188.
- Derrien, M., et al. (2019). Akkermansia muciniphila: a resident of the mucosal layer of the human gut with a potential role in health and disease. Nature Reviews Microbiology, 17(12), 727-734.
- Wang, L., et al. (2021). Pasteurised Akkermansia muciniphila improves glucose homeostasis and reduces low-grade inflammation in diet-induced obese mice. Beneficial Microbes, 12(1), 1-11.
- Geerlings, S. Y., et al. (2018). Akkermansia muciniphila as a next-generation probiotic: from lab to clinic. Critical Reviews in Microbiology, 44(5), 567-579.
- Chelakkot, C., et al. (2018). Akkermansia muciniphila-derived extracellular vesicles improve gut barrier function and ameliorate metabolic endotoxemia. Nutrients, 10(9), 1241.
- Wu, Z., et al. (2023). Clinical applications of Akkermansia muciniphila in metabolic disorders: a systematic review and meta-analysis. Obesity Reviews, 24(5), e13568.
- NIH National Institute of Diabetes and Digestive and Kidney Diseases. Insulin Resistance and Prediabetes
- American Diabetes Association. Type 2 Diabetes
- Mayo Clinic. Insulin resistance
- Harvard T.H. Chan School of Public Health. The Microbiome
- Cell Host & Microbe. Special Issue: Microbiome and Metabolism
