In the quest for metabolic health and life extension, Resveratrol has long been the undisputed king. Popularized by the “French Paradox” and groundbreaking research on Sirtuin activation, it has become a staple in millions of supplement cabinets. However, a superior challenger has emerged from the same chemical family: Pterostilbene.
The debate is no longer about which compound is more “powerful” in a petri dish, but rather which one actually reaches your cells. This deep dive focuses on the core differentiator: Bioavailability. We will explore why Pterostilbene’s unique molecular structure allows it to outperform Standard Resveratrol in human clinical applications.
1. The Molecular Stand-off: Structure Determines Destiny
Both Resveratrol and Pterostilbene are stilbenoids—natural polyphenols produced by plants as a defense mechanism against pathogens.
The Methoxy Advantage
The fundamental difference lies in their chemical “clothing.”
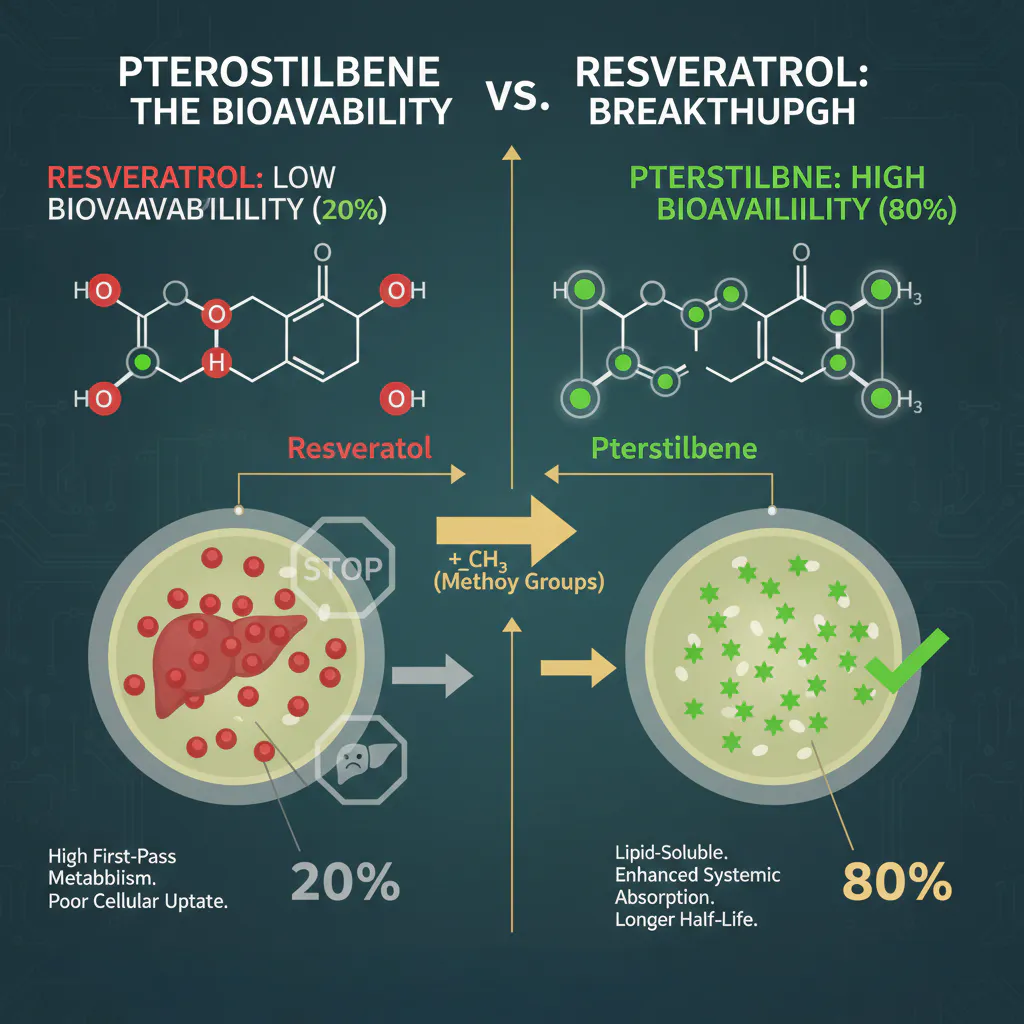
- Resveratrol (3,5,4′-trihydroxystilbene) has three hydroxyl groups. These groups are highly reactive, which sounds good for antioxidants, but it makes the molecule “sticky” and easy for the liver to capture and eliminate.
- Pterostilbene is a dimethylated version of Resveratrol. It replaces two of those hydroxyl groups with methoxy groups.
This slight structural change makes Pterostilbene significantly more lipophilic (fat-soluble). While Resveratrol struggles to pass through cell membranes, Pterostilbene slips through with ease, leading to vastly superior cellular uptake.
2. Bioavailability: The 20% vs. 80% Gap
The most cited reason to switch to a Pterostilbene vs. Resveratrol protocol is the stark contrast in oral absorption.
The Resveratrol Bioavailability Problem
Clinical studies have shown that while the human body can absorb about 70% of an oral dose of Resveratrol, its systemic bioavailability is less than 20% (and often as low as 1%). This is due to Extensive First-Pass Metabolism. The liver and intestines “conjugate” Resveratrol into sulfates and glucuronides so quickly that the “free” active form almost never reaches the brain or heart.
The Pterostilbene Solution
Research indicates that Pterostilbene has a bioavailability of approximately 80%.
- Resistance to Metabolism: Because of its methylated structure, the liver does not recognize Pterostilbene as an immediate “waste product.”
- Longer Half-Life: Pterostilbene stays in your system for roughly 105 minutes, compared to Resveratrol’s meager 14 minutes.
3. Crossing the Blood-Brain Barrier (BBB)
For users interested in Nootropics and cognitive health, Pterostilbene is the clear winner. Because it is more lipophilic, it is much more efficient at crossing the Blood-Brain Barrier.
- Neuroprotection: Pterostilbene has been shown to modulate oxidative stress in the hippocampus more effectively than Resveratrol.
- Cognitive Enhancement: By activating the PPAR-alpha receptor, Pterostilbene supports the expression of genes involved in memory and learning.
Technical Note: When evaluating raw materials, ensuring Pterostilbene (99% HPLC) purity is essential to ensure you are getting the methylated form without standard Resveratrol fillers.
4. Mechanisms of Action: Sirtuins, AMPK, and NF-kB
While the bioavailability differs, both compounds target similar longevity pathways, but with different intensities.
A. Sirtuin 1 (SIRT1) Activation
Both compounds mimic caloric restriction by activating SIRT1, the “longevity gene” that repairs DNA. However, because Pterostilbene reaches higher concentrations in the tissues, it can theoretically drive SIRT1 activation more consistently throughout the day.
B. AMPK Activation
Pterostilbene is a potent activator of AMPK (Adenosine Monophosphate-activated Protein Kinase). This enzyme regulates how the body burns fat and processes sugar.
- Product Synergy: Stacking Pterostilbene with Berberine HCl creates a powerful metabolic “one-two punch” for insulin sensitivity.
C. Inflammation and NF-kB
Pterostilbene has shown superior ability in inhibiting NF-kB, a protein complex that controls the transcription of DNA and is involved in the body’s inflammatory response. This makes it an ideal candidate for managing chronic, low-grade systemic inflammation.
5. Purity and Testing: The Importance of 99% HPLC
In the bulk extract market, “Pterostilbene” can often be diluted or poorly extracted. To ensure the 80% bioavailability mentioned in clinical trials, the product must meet the following:
- HPLC Verification: Ensures the Trans-Pterostilbene isomer is present at 99% or higher.
- Non-GMO Sourcing: High-quality Pterostilbene is often derived from blueberries or synthetic-identical processes that bypass genetic modification.
- Heavy Metal Screening: Essential for long-term daily supplementation.
6. Synergistic Stacking: Resveratrol AND Pterostilbene?
Some advanced biohackers choose not to pick just one.
- The Theory: Resveratrol may have unique benefits in the gut microbiome, while Pterostilbene handles systemic and neurological health.
- The Ratio: A common stack is a 1:1 ratio, ensuring the gut receives the benefits of Resveratrol while the bloodstream and brain receive the high-bioavailability Pterostilbene.
7. Data Overview
7.1 Bioabsorption, Utilization, and Bioactivity
| Category | Resveratrol | Pterostilbene | Key Difference / Interpretation |
| Chemical classification | Stilbene polyphenol (3,5,4′-trihydroxy-trans-stilbene) | Dimethylated stilbene (3,5-dimethoxy-4′-hydroxy-trans-stilbene) | Methoxy substitution increases lipophilicity and metabolic stability |
| Structural polarity | Higher polarity due to three hydroxyl groups | Lower polarity due to two methoxy groups | Lower polarity improves membrane permeability |
| Lipophilicity (logP) | Moderate | Higher lipophilicity | Pterostilbene crosses cell membranes more easily |
| Oral bioavailability | ~20% or lower | ~80% (animal PK studies) | Pterostilbene exhibits ~4× higher bioavailability |
| Absorption efficiency | Rapid absorption but extensive first-pass metabolism | More efficient intestinal absorption | Methoxy groups reduce conjugation metabolism |
| Plasma half-life | ~14 minutes | ~105 minutes | Pterostilbene persists ~7× longer in circulation |
| Major metabolic pathways | Rapid glucuronidation and sulfation | Mainly sulfation with slower metabolism | Pterostilbene is metabolically more stable |
| Plasma concentration after oral dosing | Lower parent compound levels | Higher circulating levels of parent compound and metabolites | Higher systemic exposure for pterostilbene |
| Cellular uptake | Limited due to polarity | High cellular uptake | Lipophilicity enhances membrane diffusion |
| Primary molecular targets | SIRT1 activation, AMPK activation, antioxidant pathways | SIRT1 activation, AMPK activation, PGC-1α signaling | Both share similar longevity pathways |
| SIRT1 activation potency | Moderate activation | Slightly stronger activation reported in some assays | May produce stronger downstream signaling |
| Mitochondrial/energy metabolism effects | Activates AMPK → improves metabolic function | Activates AMPK–SIRT1–PGC-1α axis | Pterostilbene shows strong mitochondrial protective effects |
| Antioxidant capacity | Strong antioxidant and anti-inflammatory activity | Comparable or slightly stronger antioxidant activity | Similar radical-scavenging capacity |
| Anti-aging evidence | Extensive literature (thousands of studies) | Growing but smaller evidence base | Resveratrol more extensively studied |
| Stability | Sensitive to oxidation, light, and heat | More chemically stable | Better formulation stability |
| Drug interaction potential | Can inhibit CYP enzymes | Lower CYP inhibition risk | Pterostilbene may have fewer interactions |
| Typical nutraceutical dose | 100–1000 mg/day | 50–250 mg/day | Higher potency and bioavailability allow lower dose |
7.2 Effects on NAD⁺–SIRT1 Signaling, Mitochondria, and Longevity Pathways
| Mechanistic Pathway | Resveratrol | Pterostilbene | Key Interpretation |
| SIRT1 activation | Direct and indirect activator of SIRT1; increases SIRT1 activity in multiple tissues | Also activates SIRT1; may produce stronger activation due to better intracellular accumulation | Both stimulate NAD⁺-dependent deacetylase activity |
| AMPK activation | Activates AMPK, which then enhances SIRT1 signaling | Activates AMPK and amplifies downstream mitochondrial pathways | AMPK acts upstream of SIRT1 |
| NAD⁺ metabolism | Enhances NAD⁺ levels and NAD⁺/NADH ratio via AMPK–SIRT1 signaling | Similar effect but may sustain NAD⁺ signaling longer due to longer half-life | Supports cellular energy metabolism |
| PGC-1α regulation | Activates PGC-1α through SIRT1-dependent deacetylation | Strong activation of PGC-1α and mitochondrial gene transcription | Promotes mitochondrial biogenesis |
| Mitochondrial biogenesis | Induces mitochondrial proteins and oxidative metabolism through AMPK activation | Strong stimulation of mitochondrial biogenesis via SIRT1/PGC-1α/SIRT3 axis | Important for anti-aging and metabolic resilience |
| Thermogenesis & energy expenditure | Moderate effect on metabolic rate and mitochondrial activity | Strong stimulation of thermogenesis and fat oxidation | Pterostilbene shows stronger metabolic effects in obesity models |
| Glucose metabolism | Improves insulin sensitivity and glucose uptake | Similar effects but stronger lipid-metabolism modulation | Both mimic calorie-restriction pathways |
| Lipid metabolism | Activates PPAR signaling and fatty-acid oxidation | Stronger effects on lipolysis and adipose metabolism | Often used in metabolic syndrome research |
| Oxidative stress regulation | Potent antioxidant; reduces ROS via SIRT1 and AMPK pathways | Comparable or stronger ROS reduction due to mitochondrial protection | Important in cardiovascular protection |
| Neuroprotection | Activates SIRT1/PGC-1α in neurons, reducing neuroinflammation and oxidative stress | Greater brain penetration; stronger neuroprotective potential | Potential role in Alzheimer’s and cognitive decline |
| Autophagy activation | Promotes autophagy via SIRT1–AMPK signaling | Similar mechanism; potentially stronger mitochondrial autophagy | Key longevity pathway |
| Anti-inflammatory signaling | Inhibits NF-κB and inflammatory cytokines | Comparable anti-inflammatory effect | Supports cardiometabolic health |
| Epigenetic regulation | Modulates SIRT1-dependent histone deacetylation and DNA methylation | Similar epigenetic effects but less studied | Important for gene regulation and aging |
8. Safety and Dosage
Despite its higher absorption, Pterostilbene has an excellent safety profile.
Clinical Findings: Human trials have shown that doses up to 250mg/day are safe and do not cause significant adverse effects on hepatic or renal function.
Standard Dose: 50mg to 250mg per day.
9. Conclusion: The Bioavailability Champion
While Resveratrol will always be the “famous” cousin, Pterostilbene is the “efficient” choice. For anyone serious about metabolic optimization, DNA repair, and cognitive longevity, the Pterostilbene vs. Resveratrol bioavailability debate is settled by the chemistry: Methylation equals absorption.
By choosing a high-purity, Non-GMO Pterostilbene, you ensure that your investment in your health actually reaches its cellular target.
References (Scientific Bibliography)
- Kapetanovic, I. M., et al. (2011). “Pharmacokinetics, bioavailability, and metabolic profile of resveratrol and its dimethylether analog, pterostilbene, in rats.” Cancer Chemotherapy and Pharmacology.
- Riche, D. M., et al. (2013). “Analysis of Safety from a Clinical Trial with Pterostilbene.” Journal of Toxicology.
- Chang, J., et al. (2012). “Low-dose pterostilbene, but not resveratrol, is a potent neuromodulator in aging and Alzheimer’s disease.” Neurobiology of Aging.
- McCormack, D., & McFadden, D. (2013). “A Review of Pterostilbene Antioxidant Activity and Disease Modification.” Oxidative Medicine and Cellular Longevity.
- Yeo, S. C., et al. (2013). “Pharmacokinetics and metabolic profiling of pterostilbene in rats.” Free Radical Biology and Medicine.
- Kapetanovic IM et al. Pharmacokinetics, oral bioavailability, and metabolic profile of resveratrol and pterostilbene. Cancer Chemother Pharmacol.
- Socała K et al. Dual GSK3β/SIRT1 modulators for Alzheimer’s disease. Frontiers in Pharmacology.
- McCormack & McFadden. Resveratrol vs pterostilbene bioavailability analysis.
- Rimando AM et al. Pterostilbene pharmacokinetics and bioavailability.
- Liu Y et al. Pterostilbene activates AMPK–SIRT1–PGC-1α signaling. Biochem Pharmacol.
- NOVOS Longevity Research Report. Pterostilbene vs resveratrol comparison.
